
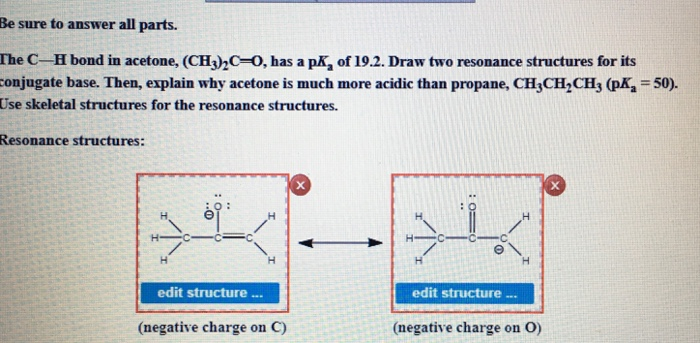
The C-H bond in acetone, (CH3_)_2C=0, has a pKa of 19.2. Draw two resonance structures for its conjugate base. | Homework.Study.com

the c―h bond in acetone, (ch3)2c═o, has a pka of 19.2. draw two resonance structures for its conjugate - brainly.com

OneClass: Draw the structure of the conjugate acid of acetone (CH3COCH3) Draw the structure of the co...

organic chemistry - Why is H2O a weaker acid than acetylacetone? Shouldn't a hydrogen connected to an oxygen be more acidic than a hydrogen connected to a carbon atom? - Chemistry Stack
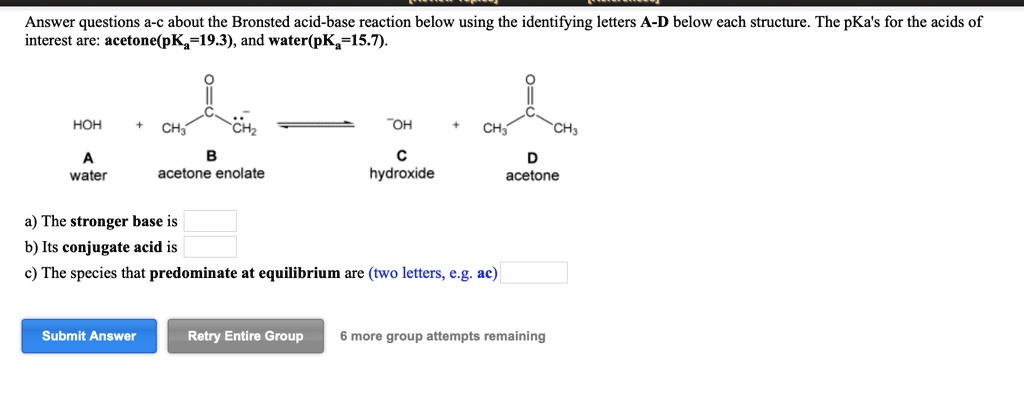
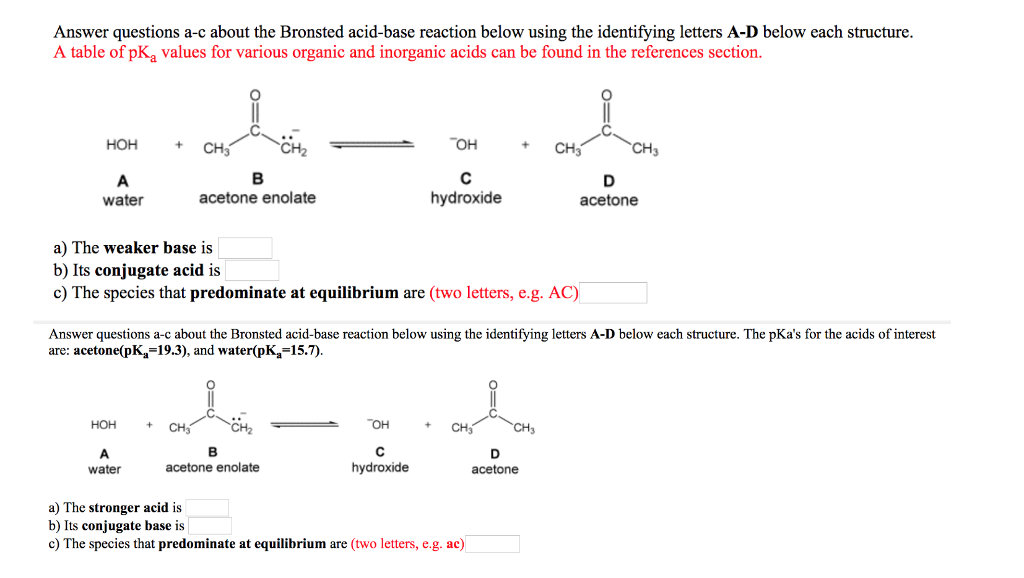
SOLVED: Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D below each structure. The pKa's for the acids of interest are: acetone (pKa -19.3) and water (pKa

2,4-Pentanedione is a considerably stronger acid than is acetone. Write a structural formula for the conjugate base of each acid and account for the greater stability of the conjugate base from 2,4-pentanedione.
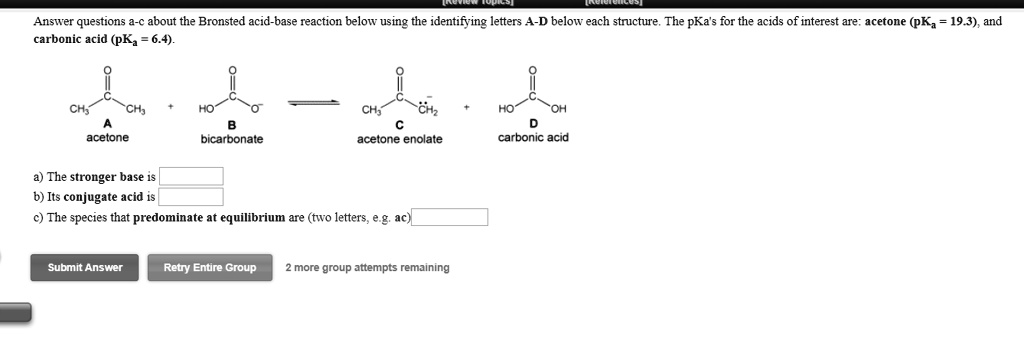
SOLVED: Answeer questions about the Bronsted acid-base reaction below using the identifying letters A-D below each stnicture, The pKa's for the acids of interest are: acetone (pK, 19.3) , and carbonic acid (


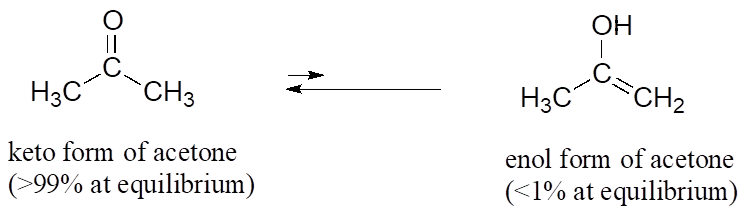


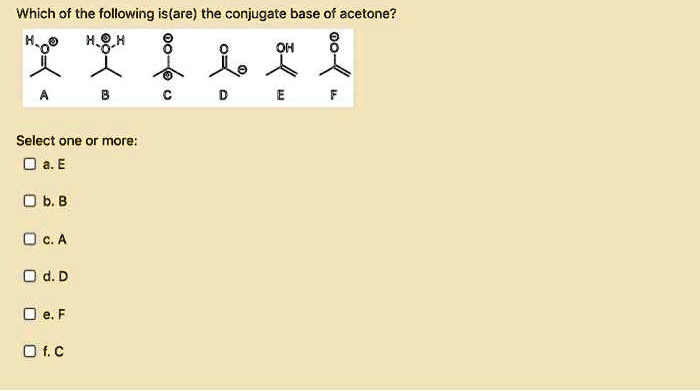






![Solved] As noted in Table 3.1, the pKa of acetone | SolutionInn Solved] As noted in Table 3.1, the pKa of acetone | SolutionInn](https://dsd5zvtm8ll6.cloudfront.net/si.question.images/image/images11/877-C-O-S(329).png)